The Mehta Research Group works primarily in India, Sub-Saharan Africa, and Latin America. Our focus is on vulnerable populations including mothers and children, and those suffering from infectious diseases such as HIV infection, Tuberculosis, Dengue virus infection, Zika virus infection, and Malaria.
Projects
Precision Nutrition for Population Health
Precision Nutrition represents a transdisciplinary approach to tackle complex nutrition and health challenges and understand the sources of inter- and intra-individual variation to diet and nutritional interventions. Dr. Mehta is the director of NIH’s first training program on Artificial Intelligence and Precision Nutrition. He is Founding Director of the Cornell Joan Klein Jacobs Center for Precision Nutrition and Health and co-lead of the NIH Research Coordinating Center (RCC) for the Nutrition for Precision Health (NPH) consortium.
Artificial Intelligence and Precision Nutrition: Our mission is to train the next generation of scientists and equip them with the skills necessary to compile, mine, analyze, and interpret big and complex datasets related to nutrition and health.
Nutrition for Precision Health: The multisite national NPH research effort will recruit 10,000 participants to discover novel diet-health linkages and develop new diet and nutritional guidelines for the prevention and mitigation of diet-related diseases. The RCC will provide expertise in design and analytics, data curation and systems, clinical trial oversight, study implementation, and will serve as data and safety monitoring site. The Cornell Jacobs Center will integrate novel approaches in precision nutrition, artificial intelligence, and technology to improve population health.
Novel Technologies
Point of Care Diagnostics for Nutrition, Infection, and Cancer
Integrating the latest advances in technology has been a common thread across our work over the last decade - including building better data systems for low resource and connectivity contexts, using biotechnology to enhance the nutritional interventions in our clinical trials, and inventing point-of-care diagnostics for nutrition - and we are now increasingly embracing artificial intelligence and machine learning to discover and solve major challenges to optimal nutrition and health.
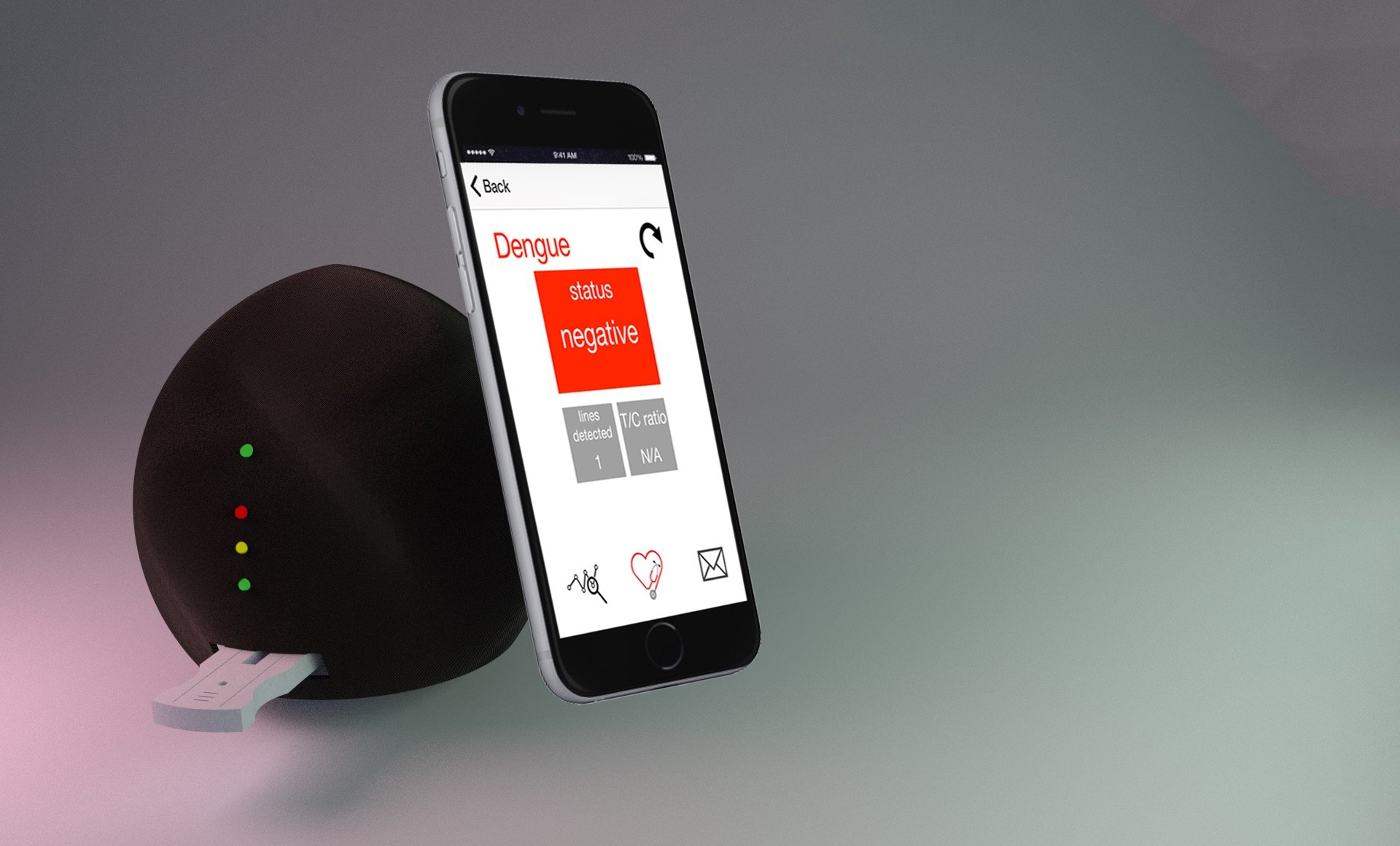
Dr. Mehta is the co-inventor of the Cornell Nutriphone, along with David Erickson, a NSF-funded mobile-based platform for real-time quantitative determination of biomarkers of nutritional status. This technology allow us to measure the status of iron and vitamins A, D, and B12 from a single drop of blood. Our work with point-of-care diagnostics has resulted in several issued patents and more than 15 publications in the last five years.
In recent work, we have shown feasibility of using salivary biomarkers to measure micronutrient status with the NutriPhone. This work resulted in a $100K prize in 2020 from the National Institutes of Health for our concept of noninvasive rapid diagnostics and the Rainer Gross Award for Innovations in Nutrition and Health in 2016.
Our team has developed and validated the Cornell FeverPhone, a NIH-funded mobile-based platform for point-of-care differential diagnosis of six common causes of acute febrile illness. The FeverPhone system builds on our team’s extensive background on the development of smartphone based diagnostics, infectious disease, and global health and enables actionable diagnosis in around 15 minutes.
We have successfully field validated these point-of-care devices for multiple infections for use in low- and middle-income settings in the US and internationally. We are now working to translate these findings through transfer of technology and scale up of production for some of these tests including those for COVID-19 in Ecuador.
MRG has two major projects in the mHealth space - ConnEDCt, a database solution for collecting data on tablets and smartphones in resource-limited settings with inconsistent internet; and a SMS-based adherence improvement project for patients with tuberculosis at our field site in India.
Infectious Disease Surveillance
Our work in this area has focused mainly on the interplay between nutrition and infection, particularly in the context of large cohort studies and active surveillance programs, focused on pregnant women and children. Dr. Mehta has led or supported several randomized clinical trials focusing on nutritional interventions in multiple high-burden settings including sub-Saharan Africa and India and with diverse populations such as adults, infants, and children, and people living with HIV and tuberculosis (TB) infection.
Our research has highlighted the potential role of vitamin D as a modifiable risk factor for slowing disease progression and disease severity both in the context of HIV and TB.
Building upon this work, Dr. Mehta is currently PI on a trial of vitamin D supplementation in patients with TB and those with HIV co-infection (https://clinicaltrials.gov/ct2/show/NCT01992263). The major outcomes of interest, in addition to treatment response, include immune competence and effect on vitamin D status. In another study, our group is collaborating with the National Institute of Nutrition in Hyderabad, India to examine the association of vitamin D with immune activation in hospitalized children. In Kenya, the research group is focusing on shedding more light on the relationship of vitamin D status with vaccine responses among infants.
In recent work, we have also highlighted how the dual epidemic of diabetes and TB is posing a major challenge for high-burden settings such as India. Even at our field site, patients with TB who also have diabetes have a three-fold higher risk of having drug resistance to the first line TB treatment. Further, current recommendations for screening for diabetes or prediabetes will miss nearly 90% of those at risk.
MRG is also helping setup active surveillance systems for infectious diseases such as Dengue, Tuberculosis and COVID-19 in Ecuador and India to facilitate early detection and triage/referral of various illnesses, interrupt transmission cycles, and to characterize the background population for future intervention, efficacy, and effectiveness studies.
Randomized Trials
Food and Biofortification-based research to improve maternal and child health
Biofortification is the process of increasing the micronutrient levels in crops through breeding. Recent advancements in this technology can improve the content of several nutrients, particularly vitamin A, iron, and zinc by several-fold, compared to conventional crops. Dr. Mehta has served as Technical Consultant on Biofortification for the World Health Organization in collaboration with the Food and Agriculture Organization.
MRG is examining the efficacy of biofortification as a sustainable agriculture-based nutritional intervention to improve health status in two large randomized trials in the urban slums of Mumbai with support from HarvestPlus/International Food Policy Research Institute. In addition, Dr. Mehta is Co-Investigator in a new trial funded by the Centers for Disease Control to examine efficacy of fortified salt among women of reproductive age, to prevent anemia and micronutrient deficiencies and improve health.
Pearl Millet (https://clinicaltrials.gov/ct2/show/NCT02233764)
A Randomized Trial of Pearl Millet in 12-18 month old Children in the urban slums of Mumbai, India
The Pearl Millet trial focuses on children 12-18 months of age and a high iron- and zinc-variety of pearl millet. This feeding trial examined the impact of biofortified pearl millet-based complementary food on iron and zinc status, growth, and immune and cognitive function.
Multiple Biofortified Food Crops Project (MBFC) (http://clinicaltrials.gov/ct2/show/NCT02648893)
A Randomized Trial of Multiple Biofortified Food Crops in 6-24 month old Children in rural and peri-urban South India
MBFC expands the crop portfolio to include orange-fleshed sweet potatoes, wheat, and lentils, in addition to pearl millet to deliver 50-70% of the estimated average requirements for vitamin A, iron, and zinc to breastfeeding mothers and their 6-12 months old infants. This feeding trial expands the crop portfolio to include orange-fleshed sweet potatoes, wheat, and lentils, in addition to pearl millet to deliver 50-70% of the estimated average requirements for vitamin A, iron, and zinc to breastfeeding mothers and their 6-12 months old infants.
Evidence Synthesis and Translation of Knowledge
Our research group is actively involved with evidence synthesis and often works with colleagues at the World Health Organization on high-priority areas to synthesize evidence and inform international guidelines related to nutrition and infection. Dr. Mehta is faculty of the WHO/Cochrane/Cornell Summer Institute for Systematic Reviews in Nutrition for Global Policy Making, and is a Cochrane affiliate at Cornell University. The Summer Institute is a collaboration of the WHO/PAHO Collaborating Center on Implementation Research in Nutrition and Global Policy in the Division of Nutritional Sciences at Cornell, the only WHO designated center for nutrition in the United States. In this role, Dr. Mehta often works with colleagues at WHO on high-priority areas to synthesize evidence to inform international guidelines related to nutrition and infection such as infant feeding in the context of SARS-CoV2, Zika virus, and Ebola virus infections. For example, we conducted the first meta-analysis and systematic review on improving iron status through biofortified food crops, and Dr. Mehta provided expert consultation to the WHO on the feasibility of such a study. We routinely conduct systematic reviews and synthesize evidence in partnership with guideline-forming agencies and implementing organizations (See Publications page). Separately, we are also conducting living systematic reviews on the transmission of SARS-CoV-2 through breastfeeding - updates can be found here.
The Institute has trained over 140 participants from 25 countries over the past eight years in the development of systematic reviews.
Nutrition, Inflammation, Microbiome and Immune Response
A vital area of focus for the Mehta Research Group is the connection between nutritional status, inflammation and the gut microbiome. Within this overall focus, a major emphasis is on vulnerable populations, particularly mothers and children. MRG has conducted randomized trials, cohort studies, and surveillance programs in the urban slums of Mumbai and southern India. Recent work has focused on elucidating changes in how the microbiome and immune function co-develop in the first two years of life and the role of nutrition in diversity and function of the gut microbiome in young infants, to inform interventions in settings with high burden of nutritional deficiencies and inflammation.
Dr. Mehta is currently Co-Investigator on a randomized trial (NCT03853304) funded by the Centers for Disease Control and Prevention, which is being conducted to examine the efficacy of quadruple fortified salt (QFS; iron, iodine, vitamin B12, folic acid) on anemia and micronutrient status among women of reproductive age over a 12-month period. This work is crucial to understanding the effects of vitamin B12 on the gut microbiome. He is also PI on two randomized efficacy trials (NCT02648893, NCT02233764) to determine the effects of delivering micronutrients through biofortified crops on nutrition and the diversity and function of the gut microbiome in women and children in India.